Boston, Massachusetts (Reuters): Boston Scientific Corp said on Thursday it was recalling its Lotus range of heart devices, citing reports of problems with the locking mechanism, sending its shares down as much as 7.4 percent.
The company also delayed the submission of its U.S. marketing application for its keenly anticipated Lotus Edge device, giving its rivals an edge in the transcatheter aortic valve replacement (TAVR) market.
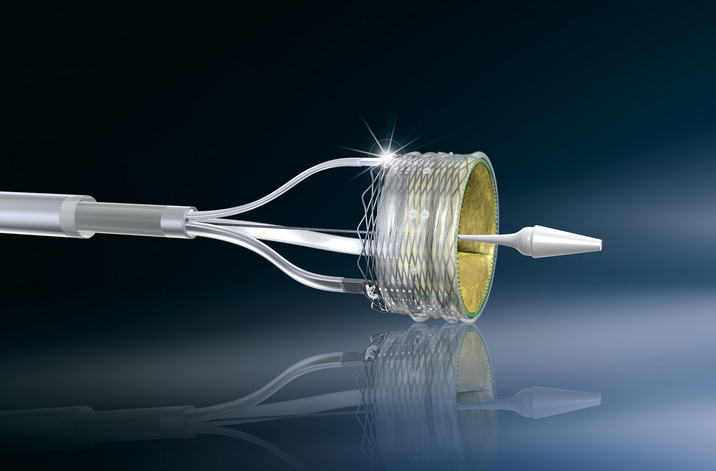
Boston Scientific's Lotus technology is used in TAVR, a minimally invasive procedure to replace a narrowed aortic valve that fails to open properly.
The TAVR market — in which Edwards Lifesciences Corp is considered the leader — could potentially exceed $5 billion by 2021, and grow 10 percent to 15 percent for the foreseeable future, Leerink analysts said in a note.


The interventional cardiology market can be unforgiving for multiple product missteps and this is perhaps the company's last chance to maintain brand equity and clinician interest, Stifel analysts said.
Boston Scientific's Lotus devices are expected to return to the European market and other regions in the fourth quarter, it said, noting the issue was caused by excess tension in the pin mechanism that was introduced during manufacturing.
The Marlborough, Massachusetts-based company had suspended implants of its next-generation Lotus Edge device in Europe last October over similar concerns, but said last month it had found a fix for the issue.
Boston Scientific on Thursday also pushed the submission of its marketing application for the device in the United States to the fourth quarter, and said it now expects to launch it in mid-2018.
It had previously expected to submit the application in May and get an approval by the end of 2017.
Analysts had previously forecast $100 million to $125 million in global sales for its Lotus devices in 2017.
Boston Scientific said it is not updating its full-year forecast, but has lowered its structural heart revenue expectations by $50 million, which translates into a 2 cent impact on its earnings per share, Evercore ISI's Vijay Kumar said.
Shares of Edwards Lifesciences rose as much as 8.4 percent to $99.99, while another rival Medtronic Plc's stock inched up about 1 percent to $80.41.
The competitive threat has been pushed back 6 to 9 months and the short-term Lotus withdrawal will obviously benefit Edward's 2017 revenues and earnings, J.P. Morgan analysts said.


